FDA Issues Recall for Popular ADHD Drug Amid Health Risk Warnings
Impact of the Nationwide Drug Recall on ADHD Patients
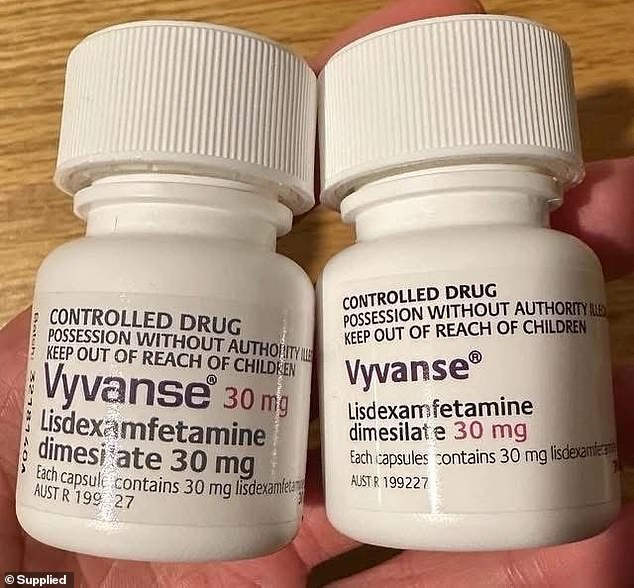
Millions of Americans who rely on medication to manage attention deficit hyperactivity disorder (ADHD) may be affected by a recent nationwide drug recall. The recall involves several lots of lisdexamfetamine dimesylate capsules, which are commonly known as Vyvanse and Arynta. These medications are prescribed for patients aged six and older to help manage symptoms of ADHD.
The recall was initiated by Sun Pharmaceutical Industries, a major pharmaceutical company, and is classified as a Class II recall by the FDA. This classification indicates that while the use of the product may cause temporary or medically reversible adverse health consequences, the risk of serious harm is considered remote.
The recall was prompted by a failure in laboratory testing, as the affected lots of medication did not dissolve properly during quality control tests. This defect could affect how the drug is absorbed in the body, potentially leading to patients receiving a lower dose than prescribed. As a result, the therapeutic effectiveness of the medication may be reduced, which could worsen ADHD symptoms and trigger fatigue or concentration problems.
Rising Prescriptions and Increased Awareness
Prescriptions for lisdexamfetamine dimesylate have seen a significant increase in the US over recent years. Data shows that from 2012 to 2023, overall stimulant prescriptions grew by about 60 percent. In 2023 alone, lisdexamfetamine accounted for roughly 19 percent of all stimulant prescriptions, with more than nine million prescriptions dispensed.
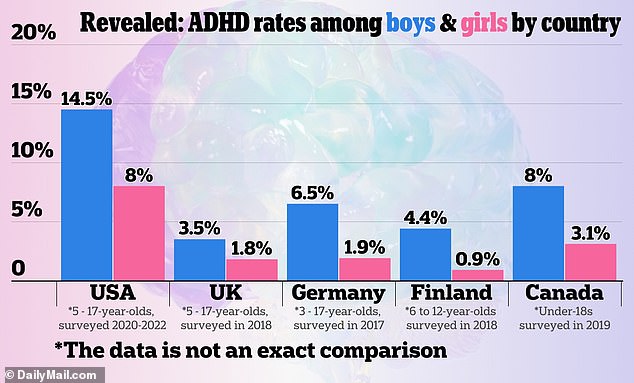
This increase coincides with a rise in ADHD diagnoses, particularly among adults and women, as awareness and screening have improved. The growth of telehealth services has also made it easier for people to access ADHD evaluations and prescriptions.
Details of the Affected Batches
The recall covers 100-count bottles of 10 mg to 70 mg capsules with expiration dates ranging from February 2026 to May 2026. The capsules were manufactured by Ohm Laboratories, a subsidiary of Sun Pharmaceutical Industries, based in New Brunswick, New Jersey.
Patients who believe they may have medication from an affected lot are advised not to stop taking it without consulting a healthcare provider. The FDA urges patients to contact their doctor or pharmacist for further guidance and to arrange for a safe replacement if necessary. For more information, patients can visit the FDA's official recall page or contact Sun Pharmaceutical Industries directly.
Understanding ADHD Medications
Around 22 million Americans are estimated to have ADHD, and just over half of these individuals are prescribed medication to manage their symptoms, which can include impulsiveness, disorganization, and difficulty focusing. Medications are broadly classified into two main categories: stimulants and non-stimulants.
Stimulants, the most common type used to treat ADHD, include methylphenidate and amphetamine-based medications like lisdexamfetamine dimesylate. These medications improve the transmission of dopamine, a brain chemical that affects mood, motivation, and movement. Non-stimulant options such as atomoxetine, clonidine, and guanfacine can also be used if stimulants are not effective or well tolerated. These help improve the transmission of norepinephrine, a hormone that aids in alertness and focus.
Lisdexamfetamine dimesylate is a prodrug, meaning it becomes active only after being metabolized in the body into dextroamphetamine. This design helps provide a smoother, longer-lasting effect and reduces the potential for misuse compared to some other stimulants.
Some of the best-known brand names for ADHD medications include Adderall, Ritalin, Vyvanse, Focalin, Concerta, and Daytrana.
Recalled Batches
The affected batches include:
- 10 mg: AD42468 (exp. 2/28/2026) and AD48705 (exp. 4/30/2026)
- 20 mg: AD42469 (exp. 2/28/2026) and AD48707 (exp. 4/30/2026)
- 30 mg: AD42470 (exp. 2/28/2026) and AD48708 (exp. 4/30/2026)
- 40 mg: AD48709 (exp. 4/30/2026) and AD50894 (exp. 5/31/2026)
- 50 mg: AD48710 (exp. 4/30/2026) and AD50895 (exp. 5/31/2026)
- 60 mg: AD48711 (exp. 4/30/2026) and AD50896 (exp. 5/31/2026)
- 70 mg: AD48712 (exp. 4/30/2026) and AD50898 (exp. 5/31/2026)