Baby Formula Recall Widens as More Infants Sicken from Deadly Bacteria
Expanding Recall of Infant Formula Due to Botulism Concerns
A baby formula brand has expanded its recall of infant formula after a dangerous bacteria was found, leading to multiple hospitalizations across the country. The company, ByHeart, based in Reading, Pennsylvania, is now recalling all of its products following an investigation by the FDA into a recent outbreak of infant botulism.
The FDA notified ByHeart on November 7 about 83 reported cases of infant botulism nationwide since August 2025. The number of cases has now risen to 84. To date, 15 infants have been hospitalized after consuming ByHeart formula.
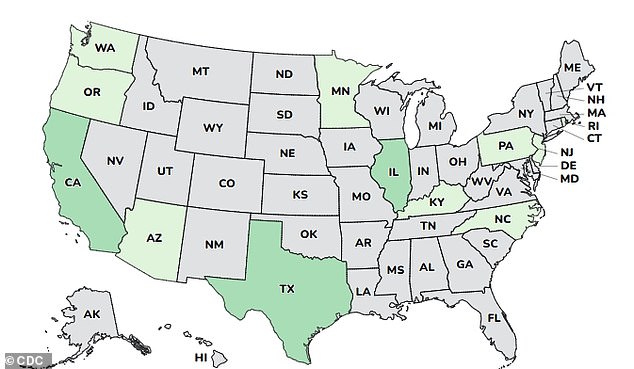
The affected cases were reported in 12 states: Arizona, California, Illinois, Minnesota, New Jersey, Oregon, Pennsylvania, Rhode Island, Kentucky, North Carolina, Texas, and Washington.
Although no direct connection has been confirmed between any infant formula brand and the illness, health officials are concerned that other lots of ByHeart Whole Nutrition infant formula may be contaminated.


The California Department of Public Health tested a can of ByHeart powdered infant formula that was fed to an infant with infant botulism. Preliminary results suggest the presence of the bacteria that produce botulinum toxin.
The voluntary recall applies to all lot numbers and all sizes of cans and single serve packets of ByHeart infant formula.

The CDC advises anyone in possession of the recalled formula to record the lot number and "best by" date if possible. Consumers who still have leftover powdered formula should store it safely for at least one month rather than discarding it immediately. This precaution allows time for health officials to collect samples if an infant develops symptoms consistent with infant botulism, such as poor feeding, constipation, or muscle weakness.
If no symptoms occur after a month, the leftover formula should be discarded. Parents are also urged to clean and disinfect any items or surfaces that came into contact with the recalled formula using hot, soapy water or a dishwasher to reduce the risk of contamination.

The CDC emphasized that these measures are part of a broader effort to support the FDA's ongoing investigation and protect infant health while the source of the outbreak is being examined.
Understanding Infant Botulism
Infant botulism is a rare but potentially fatal condition that affects babies, usually under 12 months old. In the US, there are typically around 200 to 300 cases of infant botulism reported each year. The majority of these, roughly two-thirds, are infant botulism, which usually affects babies under one year old.
It happens when spores of the bacterium Clostridium botulinum enter an infant's intestines, where they can grow and produce botulinum toxin—one of the most potent natural toxins known.
Symptoms can include constipation, poor feeding, drooping eyelids, weak cry, low muscle tone, and in severe cases, respiratory difficulty or arrest. In infant botulism, the food doesn't contain toxin; it contains spores that have the potential to produce toxin inside the baby's body.
The best-known food linked to infant botulism is honey, and health authorities advise never giving honey to babies under 12 months. Occasionally, spores can also be found in dusty home environments, unwashed produce, or powdered foods, though these are much rarer routes of exposure.
The main treatment for infant botulism is an antitoxin called Botulism Immune Globulin Intravenous (Human), or BIG-IV, administered via a single intravenous infusion. Supportive care is also essential, which may include hospitalization, breathing support with a ventilator if needed, and IV fluids or tube feedings to maintain nutrition if the infant has difficulty swallowing.
Early diagnosis and treatment are crucial for a favorable outcome. While death is rare at less than one percent, recovery can be a lengthy process, requiring months or even years for full recovery. To date, no deaths have been reported from the outbreak.
Company Response and Consumer Actions
ByHeart highlighted that the recall is being conducted out of an abundance of caution, as the FDA has not established any direct link between infant formula and the reported cases of botulism. There is also no historical precedent of infant formula causing infant botulism.
"No ByHeart product has tested positive for any contaminants," said Mia Funt, Co-Founder and President of ByHeart. "The safety and well-being of every infant who uses our formula is our absolute highest priority."
"While there has been no confirmation of contamination, this voluntary recall reflects our deep commitment to transparency and safety for babies and their parents."
Funt added that ByHeart follows the highest global and US safety standards and testing protocols for its products. Laboratory confirmation for some cases is ongoing.
Consumers who purchased products from the affected batches have been advised to immediately discontinue use and dispose of the formula. ByHeart will replace any recalled cans at no cost. Parents should contact their healthcare provider if their infant shows symptoms consistent with botulism.
Meanwhile, businesses have been instructed to stop selling the recalled ByHeart Whole Nutrition infant formula and to sanitize any surfaces that may have touched the product.